
The reaction with the unsubstituted reagents, i.e., butadiene and ethylene, takes place at moderate temperatures, the reagents must be heated and placed at a certain pressure, but the reaction takes place with activation energies that are compatible with the state correlation diagram. This reaction has the following experimental characteristics: Main characteristics of the Diels-Alder reaction
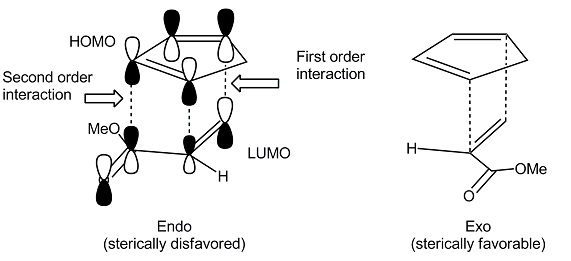
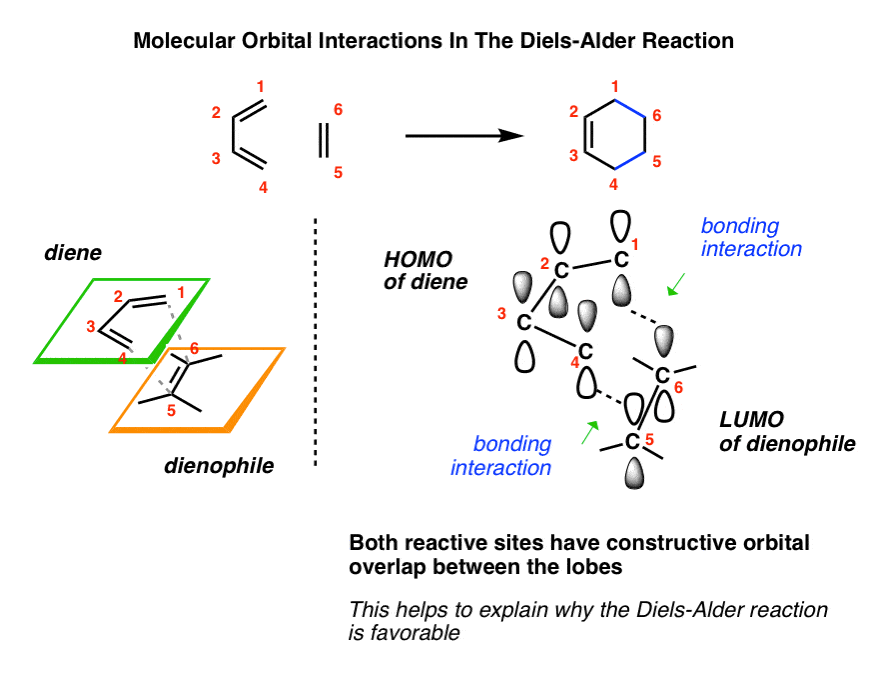
Let's take a look at its reaction mechanism and try to explain why the most significant features of this reaction occur. The diene and dienophile react on the same side of both molecular planes (suprafacially) as shown in the figure below: The diene contributes 4 π electrons and the dienophile 2 (π 4+π 2) as shown in the figure. The simplest case is the reaction between buta-1,3-diene and ethene. It is a reaction in which a conjugated diene combines with a so-called dienophile, which can be an alkene or alkyne, to give a 6-membered cyclic adduct. The Diels-Alder (DA) reaction is the pericyclic reaction par excellence, also called cycloaddition. References What is the Diels-Alder reaction?.Main reaction characteristics: HOMO-LUMO interactions.How do the substituents affect the reaction?.Main characteristics of the Diels-Alder reaction.Bibliographic Sources in Organic Chemistry.Correlation diagrams in pericyclic reactions.Named organic reactions and chemical reagents.Structure and representation of organic molecules.Identification of Organic Compounds by Spectroscopy.Heating and cooling of chemical reactions.Measurement of the melting point of a solid.Volume measuring for liquids or solutions.Substances with risk of fire and explosion.GHS system for classification and labeling of chemicals.The above also applies to the dieneophile. I'm afraid I don't know the US equivalent of this book(s) though. If you're looking for some literature though I'd recommend Clayden's Organic chemistry textbook, as it explains this a lot better than I probably am with words alone. This is why adding a Lewis acid to a Diels Alder leads to better yields, lower reaction temperatures and more regiospecificity.īut my advice is always draw the orbitals out for the diene and dieneophile, work out the energies of them by the number of nodes present. This is because the orbital coefficients are changed by the presence of these groups, and maximal orbital overlap is always most likely to form the bonds you want. You can then match the partial positives to the partial negatives on the dieneophile and vice versa. This gives you the 4 relevant orbitals and their relative energies on the diene, you then fill them up and you can easily see which orbitals are the HOMO and LUMO.įor dienes which have electron donating groups attached to them, you can draw resonance structures to see which atoms would have a partial positive an/or negative charge. For 1,3-butadiene as an example you can assign it as 4 electron pi-system, so if you draw the p-orbitals on each carbon atom on the diene and then draw out the orbitals on each carbon atom, you can tell which ones are filled first by minimising the number of nodes in each energy level and increasing from there.
